Dosimetry that runs —
so your trial does too.
From pre-clinical scaling to First-in-Human, through early and pivotal phases — protocol design, site qualification, central analysis, submission reporting. So when patient data comes in and clinical decisions have to be made, the dosimetry is ready.
The full analysis. The right advice. On time.
For novel targets and new radiopharmaceuticals, we advise on organs-at-risk that may not be established in existing literature — drawing on our dosimetric and clinical experience to inform protocol design before the first patient is dosed. Every analysis goes through a structured quality control process covering imaging and treatment data, with peer review before any report is issued.
Reports are structured for regulatory review from the start — built alongside current FDA and EMA expectations around mandated dosimetry in trials. Turnaround is fast enough to feed results into Safety Monitoring Committee cycles, so dosimetry data is available when decisions are being made, not after them.
Consistent data across every site.
By design, not by chance.
Multi-site dosimetry fails when sites use different acquisition parameters. We eliminate that problem before the trial starts — designing scanner-specific protocols, writing equipment SOPs, and qualifying each site against the same phantom-based standard.

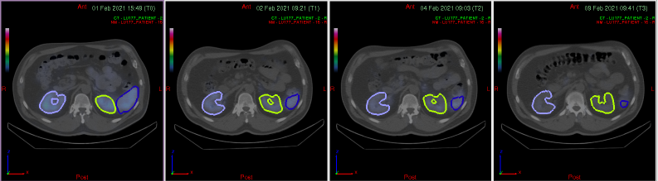
Hybrid, single-timepoint and multi-timepoint protocols tailored to nuclide and clinical constraints — collimator selection, energy windows, reconstruction parameters, calibration requirements.
Radionuclide calibrators, gamma counters, well counters, phantom validation — unified across all sites. Two scanners, two SOPs, both validated independently.
Eligibility checks, setup support, and remote or on-site training. Each site is qualified before first patient. Sites are trained on the protocol — not just handed it.
The equipment a site needs — without the procurement overhead.
Sites that lack specialist equipment for phantom studies and quantification validation can draw on our managed rental service. We handle logistics, insurance, and on-time delivery.
Pre-clinical dosimetry that holds up when it reaches the clinic.
From imaging protocol selection to absorbed dose scaling across species — pre-clinical dosimetry defines the assumptions that carry forward into first-in-human. Getting it wrong early is expensive. We have published on the methodology, not just applied it.
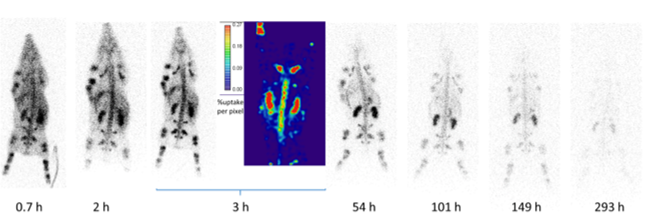
Imaging protocol selection in pre-clinical studies is not a technical detail — it directly determines the quality of the absorbed dose data that will inform first-in-human dose selection. A pragmatic but under-specified imaging schedule can double your TIAC estimate and the absorbed dose derived from it.
We also have direct experience with the scaling step itself — translating absorbed dose data across species using allometric and OLINDA/EXM-based methods. Choosing the right scaling approach for a novel radiopharmaceutical is not a formality. It requires understanding how the compound behaves in the animal model and what assumptions carry forward into the human dose estimate. This is the kind of methodological grounding we bring into pre-clinical dosimetry work.
Most dosimetry conversations start with
a single trial or a single nuclide.
Start there. We will tell you whether what we do fits what you need — and what it would take to make it work. No commitment required from that first conversation.